3
• Decline in visual acuity*
• Worsening of dry eye symptoms*
• Increased discomfort or pain of ocular surface (grittiness,
foreign body sensation, etc.)
• Discomfort or pain of eyelids or orbit*
There is a potential risk of thermal injury to eye or eyelid based
onthe device design.
2.6 FOR PRESCRIPTION USE ONLY
Federal (USA) law restricts this device to sale, distribution, or use by
or on the order of a physician. Physician training is required prior to
use of the TearCare System.
2.7 CLINICAL STUDY SUMMARY
A prospective, multicenter, randomized, non-inferiority, masked,
controlled clinical trial (“OLYMPIA”) was performed to demonstrate
the safety and eectiveness of a single TearCare System treatment
compared to a single LipiFlow Thermal Pulsation System to treat
adult patients with Meibomian Gland Dysfunction (MGD).
Study Design
This study was a prospective, randomized, single-masked,
multi-center, non-inferiority, non-significant risk device study.
Randomized subjects were followed for one month succeeding
treatment with follow-up data collected at Day 1, Week 2, and
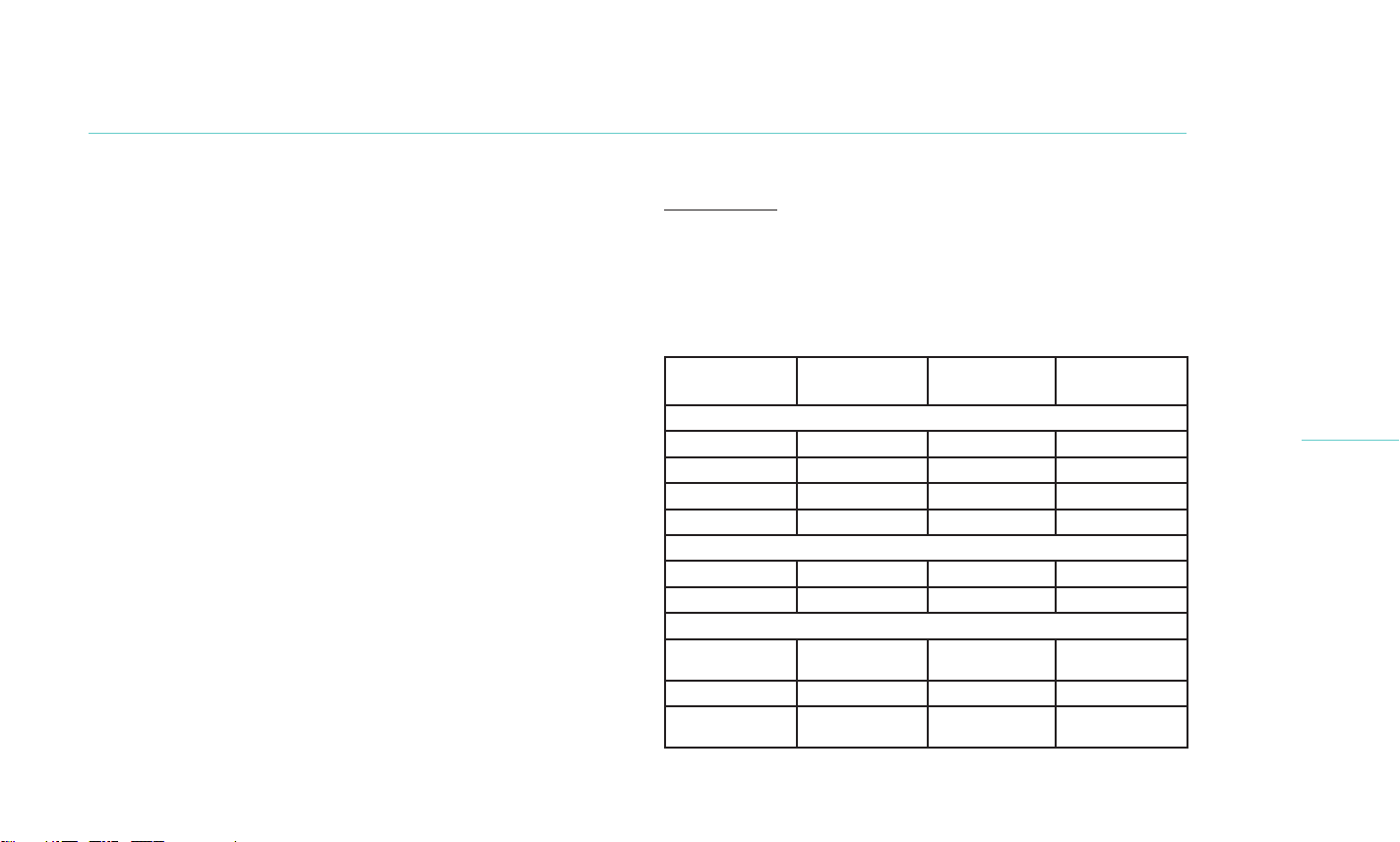
1 Month. A total of 235 subjects (470 eyes) from 10 investigative
centers in the United States participated in the study, comprised
of 169 female and 66 males, ages 22 to 91 years (mean = 55.9 ±
14.4 years). Subjects were randomized 1:1 to receive either a single
TearCare System or LipiFlow System treatment. The TearCare
treatment arm consisted of a 15-minute thermal procedure
followed immediately (i.e., within 3 minutes) by manual expression
of the meibomian glands using the Clearance Assistant. Study
subjects were grouped into two cohorts to account for a SmartLid
design change made during the study. There were 93 subjects
in Cohort 1, comprised of 47 LipiFlow and 46 TearCare subjects
treated with the prior SmartLid design. There were 142 subjects
in Cohort 2, comprised of 73 LipiFlow and 69 TearCare subjects
treated with the current SmartLid design. The eectiveness
endpoints were assessed using data from Cohort 2 and the safety
endpoints were evaluated separately for Cohort 1 and 2. The study
procedures took place between March 2019 and February 2020.
Study Endpoints
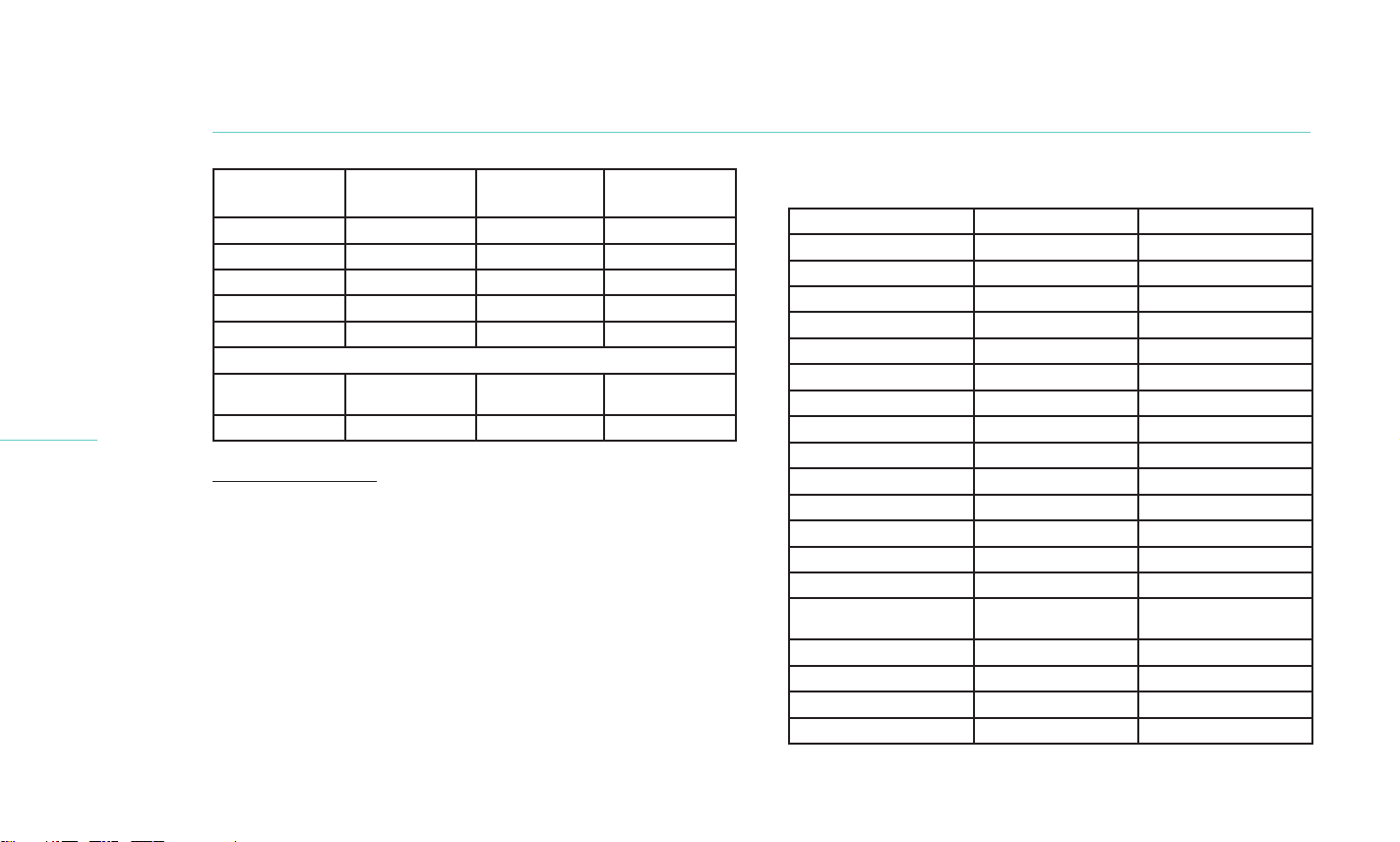
The primary eectiveness endpoints included the mean change
from baseline to 1-month in Tear Break-Up Time (TBUT) and
Total Meibomian Gland Secretion Score (MGSS). Secondary
eectiveness endpoints included the mean change from baseline
to 1-month in Ocular Surface Disease Index (OSDI) score, corneal
and conjunctival staining scores, Symptom Assessment in Dry Eye
(SANDE) scores, Eye Dryness Score and meibomian gland health.
The primary safety endpoint was the incidence of ocular adverse
events. The secondary safety endpoints included discomfort/pain
during and after the procedure, change in Best Corrected Visual
Acuity (BCVA), and change in intraocular pressure (IOP).
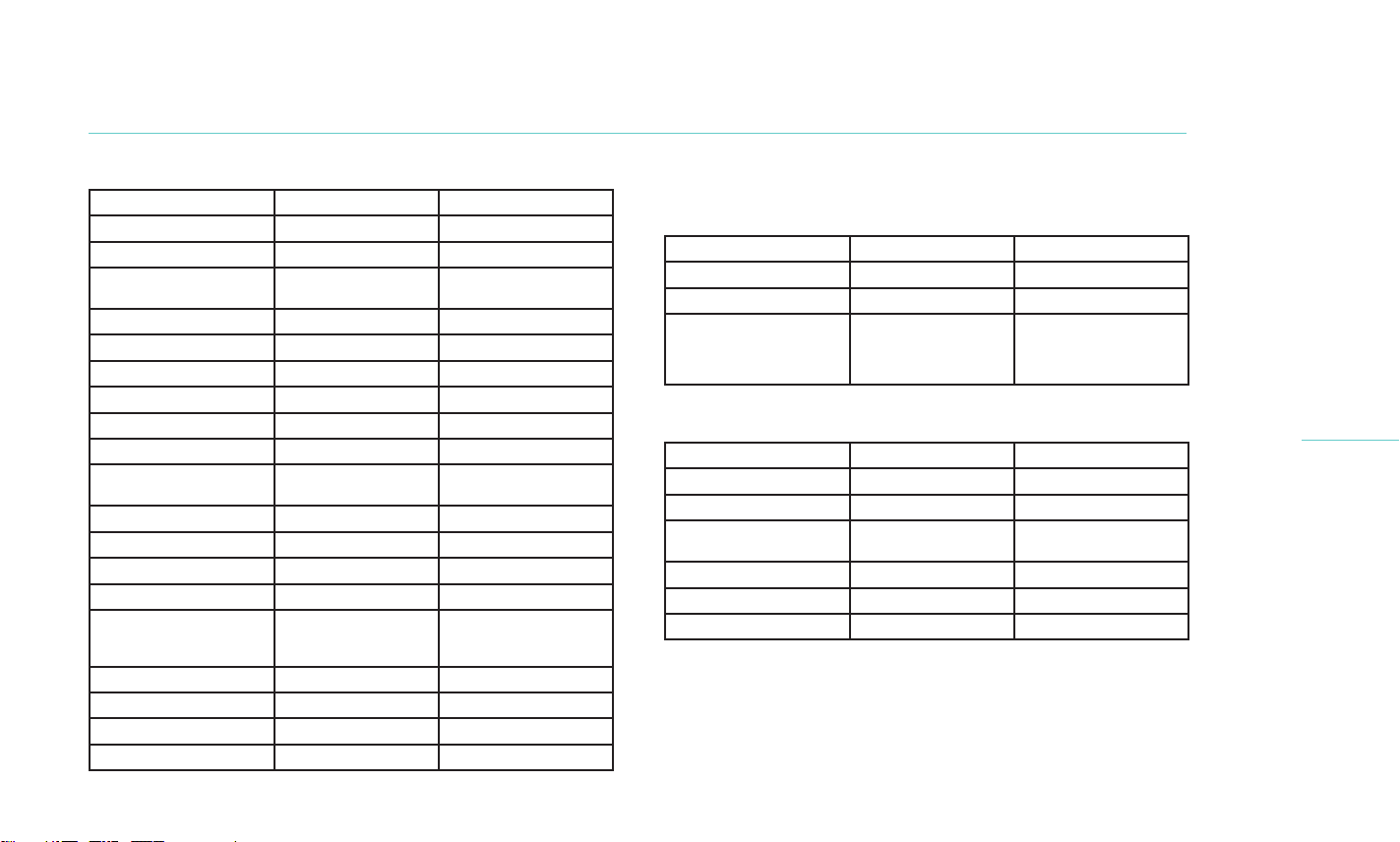
Description of Study Patients
To participate in the study, subjects were required to be at least
22 years of age with symptoms of dry eye in the past 3 months,
regularly reported use of artificial tears or lubricants over the
2. TEARCARE SYSTEM INFORMATION